Some organizations taking advantage of LifeOmic's platform.





Some organizations taking advantage of LifeOmic's platform.






We Work With Your Integrations
Don’t get rid of what you have. We have recognized integrations:
- EMRs: Epic & Cerner
- Omics data
- Public & private databases
- Wearables: Fitbit, Apple Healthkit, GoogleFit, Garmin, Oura, KetoMojo
Plus, open APIs to integrate with anything else.
We are Focused on Standards
- HL7 FHIR
- Global Alliance for Genomics & Health (GA4GH)
- Compliance: HITRUST, HIPAA & GDPR compliant


We are Focused on Standards
- HL7 FHIR
- Global Alliance for Genomics & Health (GA4GH)
- Compliance: HITRUST, HIPAA & GDPR compliant
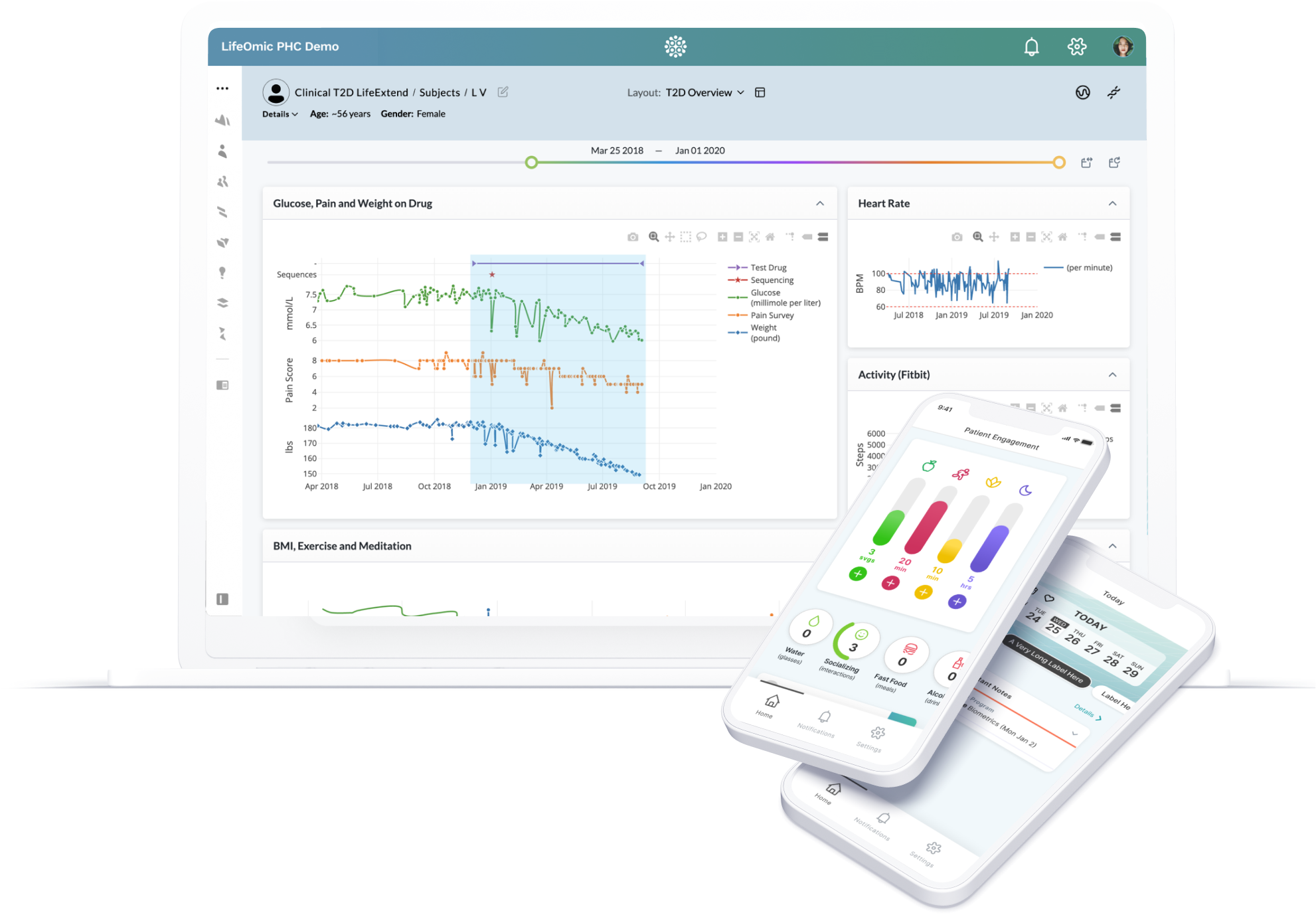
We Go FarBeyond Storage
Our data is actionable for systems, providers and patients.
- Attribute-based access controls
- Open restful APIs
- User-friendly web interface
- Real-time data analysis & visualization
- Patient engagement mobile app

LifeOmic Wins “Best Patient Data Security Solution” in MedTech Breakthrough Award Program
We believe every patient deserves to know that their sensitive health information is being handled with the utmost care and protection.
Utilize One of Our Pre-Built Solutionsor Customize a Health Data Solution

Oncology Departments
Design individualized patient treatment plans by exploring genomic data EHRs, clinical trial matching databases, wearable devices and patient-reported outcomes.
Cardiology Departments
Enable effective intervention and treatment of heart disease by analyzing EMR, imaging, device, lab, sequencing and patented-reported data in one centralized place.
Medical Device Companies
Securely collect, store and analyze diagnostic data from your medical devices. Improve post-market surveillance, AI-supported innovation and patient care.
Academic Researchers
Accelerate academic research by taking advantage of simplified data integration, Optical Character Recognition technology and machine learning-supported analysis.
Pharmaceutical Researchers
Perform big data analysis more effectively to develop impactful treatments sooner. Efficiently recruit diverse, representative clinical trial participants to speed up regulatory approvals.
Patient Care Teams
Deliver custom patient care pathways with a configurable mobile app. Enable MyChart portals, telehealth, digital treatment binders and behavior tracking.
Note: All data displayed on this website is simulated.

